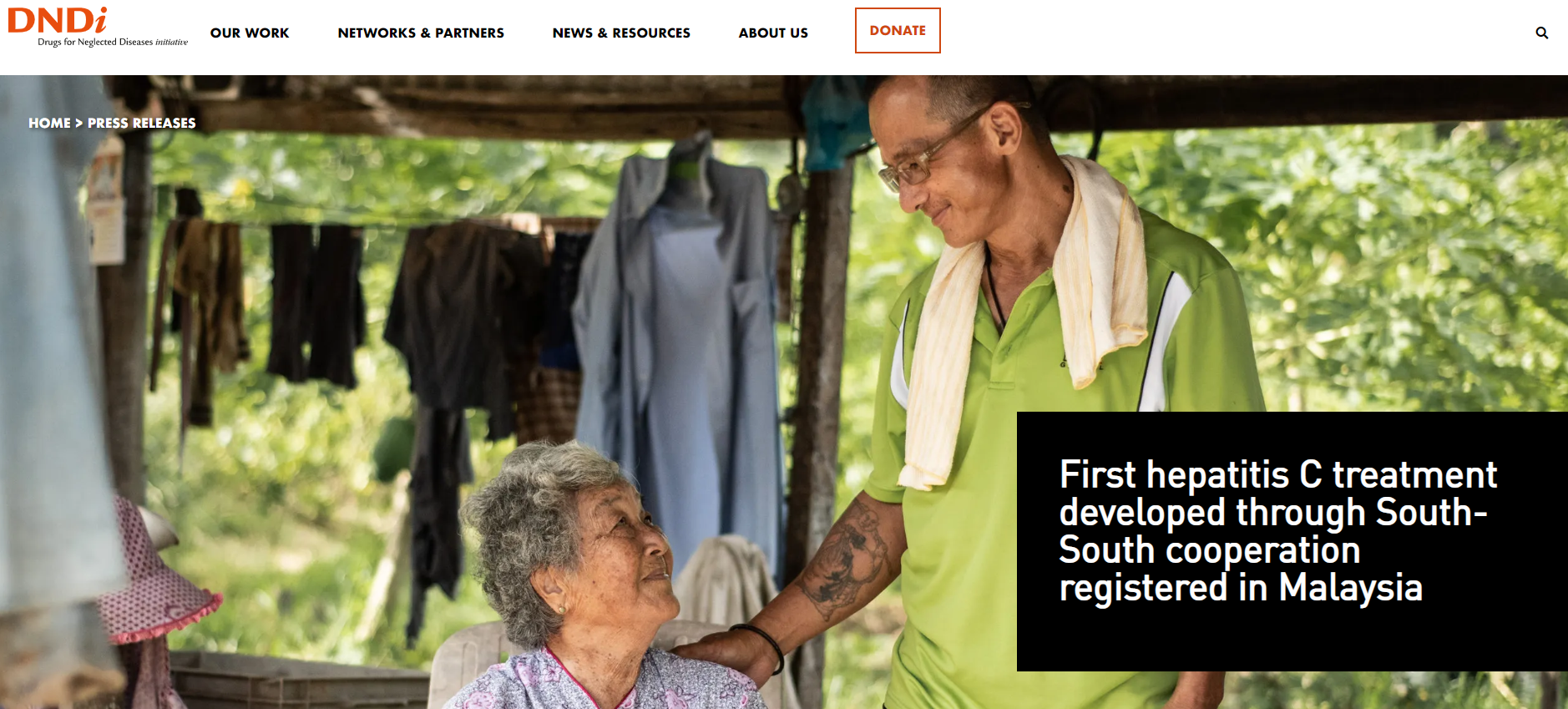
On March 8th Doppel Pharma CDMO delivered the first validation & Commercial batches of Ravidasvir, to be commercialized in Malaysia for the treatment of Hepatitis C.
Hepatitis C is a viral infection that causes liver inflammation, sometimes leading to serious liver damage including cancer.
Ravidasvir is an oral NS5A inhibitor discovered, owned and patented by Presidio Pharmaceuticals, USA. It was licensed to Egyptian drug manufacturer Pharco Pharmaceuticals and DNDi for clinical development and commercialization.
The collaboration regarding formulation and production started back in October 2017 between Doppel Pharma CDMO and Pharco Pharmaceuticals, and more broadly it consists of a partnership including the NGOs Drugs for Neglected Diseases initiative (DNDi), Médecins Sans Frontières/Doctors Without Borders (MSF), and the Malaysian pharmaceutical company Pharmaniaga Berhad as final distributor.
Currently the partnership is tirelessly working to make Ravidasvir available in other territories in Asia, Africa and Latin America.
“At Doppel we are tremendously proud of this collaboration, especially since it is enabling access to life saving medicines at affordable prices to patients all around the world“ Giorgio Oberrauch Doppel Pharma CDMO CEO said, “This achievement marks and important milestone for Doppel Pharma CDMO, and gives us confidence and strength to continue working with resiliency during these difficult and challenging times”
#theproject https://lnkd.in/eEzkdVZb
#Pharco https://lnkd.in/eSxQvAq7
#Doppel https://www.linkedin.com/posts/doppel-farmaceutici-srl_first-hepatitis-c-treatment-developed-through-activity-6916680794795069440-GaoB?utm_source=linkedin_share&utm_medium=member_desktop_web